
Welcome India Regional Visitors | Visit our Global Site
Delivering on Your EHS, Sustainability & ESG Reporting Needs With One Unified Digital Platform
For over two decades, our intuitive applications have enabled cross-functional performance excellence.
Explore our Solutions

Environment, Health & Safety (EHS)

Sustainability / Climate

ESG Reporting

Quality

Equipment & Operational Risk

Stewardship

Supply Chain Risk
A Platform Designed to Enable Success
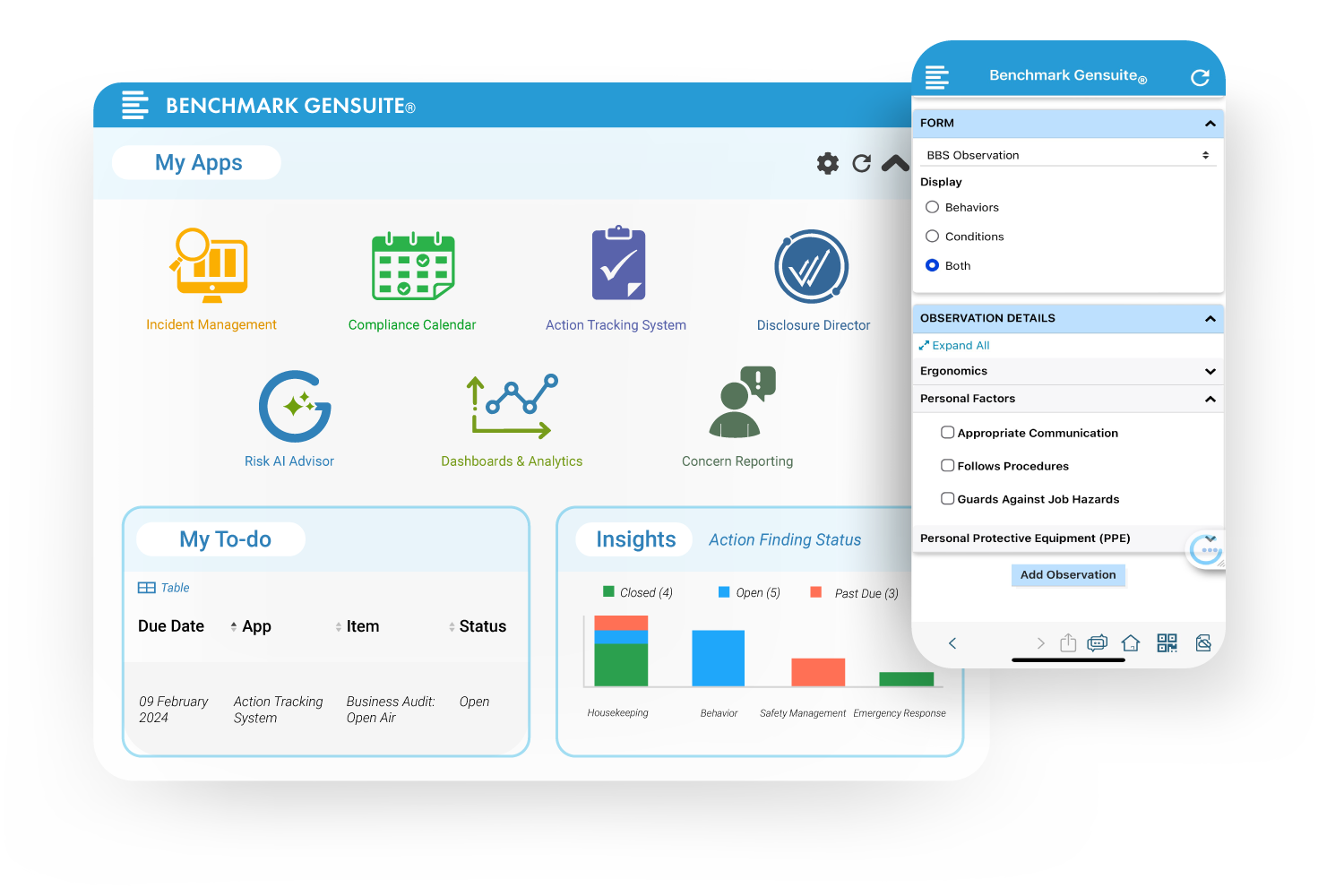
AI

Best Al in EHS
Empower data collection, user guidance, and risk & safety insights.
Analytics

Real time data
Reveal data-driven insights to drive change and executive engagement.
Mobile App
4.5 star rating
Join our 250,000+ monthly users and tap into your full solution suite from anywhere!

Proven, flexible, and powerful. Ready to deploy in 3 months or less.
Organically built by, with, and for functional leaders like you.











"The Benchmark Team is really unique. I feel very heard. You are really listening to us and actually coming back with solutions"

Sandra Neale
Global EHS Leader
"Costs for Benchmark Gensuite have been in full alignment with our expectations... There have been no hidden costs or surprises with budgets/forecasts."

John Novotny
Director, Global Environment & Sustainability
"ESG considerations enhance our company by reducing investor risk and enhancing value. It creates a greater level of resilience and output so we can outperform our competitors."

Herman Baker
Global EHS & Sustainability Director
What Makes Us Unique
Unified Platform
and IT Integrations

Cloud-Based
and Flexible

Collaborative Innovation
and Workgroups
Serving a Global Community of EHS and ESG Leaders
With over 25 years of collaborative innovation and partnership with our subscribers, our story continues to unfold.
Benchmark Gensuite® is a Certified and Accredited Partner and Service Provider of These Global Frameworks and Standard Organizations
Get started with Benchmark Gensuite.
Let us show you how we're transforming operations, workflows, and programs for millions of users— just like you— across the globe.




